Turnover Rates and Workforce Friction: Threats and Opportunities for Digital Health Companies
Why workforce issues impact digital health adoption and revenue
I’ve enjoyed a front-row seat to both the travel nurse and digital health markets over the past four years. During the steep curves of COVID-19 incidence, I witnessed the impact of healthcare workforce issues on hospitals, clinics, and physicians through the customers of our tech-enabled, remote patient monitoring (RPM) services business, Avenue Health. At the same time, my access to one of the largest employment data sets for travel nurses and contingent clinical labor in the U.S. provided a daily glimpse into the state of the healthcare workforce.
Hospitals and other care delivery organizations turn to the contingent labor market when they are unable to hire full-time staff to fill roles given a certain patient demand. In other words, they use “temps.” Full-time clinicians are burning out and quitting at high rates, thus driving demand for contingent clinician labor. The healthcare staffing industry is a multi-billion dollar market and is the largest manifestation of the friction, burnout, and willingness to quit expressed by healthcare professionals and clinicians.
At Avenue Health, our hospital and clinic partners have staff turnover problems that can affect the culture of innovation, while prospective clinic customers often cite internal workforce struggles and missing roles as reasons to delay the implementation of our digital health-based clinical model. Thus, the challenges in the healthcare workforce have direct business implications for healthcare delivery organizations themselves and the digital health companies that sell to them or rely on clinicians for adoption. Workforce issues are often overlooked by digital health companies, in particular, as the focus is primarily on clinical patient impact. Looking to solve for better health, digital health companies primarily focus on clinical evidence and medical outcomes, but lurking behind the R&D efforts and product development are both workforce barriers and opportunities that will make or break future revenue growth.
For investors, healthcare providers, consultants, and digital health companies; understanding a company’s impact on healthcare workers and other key factors is critical to picking and developing winning companies in the healthcare services market. The following article will discuss this concept in more detail and provide a conceptual model.
Turnover, Retention, and Healthcare Workforce Friction
Turnover among clinicians is a billion-dollar problem for hospitals, medical practices, and nursing homes. It is also the single biggest opportunity, barrier, and overall threat to digital transformation in U.S. healthcare.
Take, for example, a small medical practice with two medical assistants, one registered nurse, a nurse practitioner, and a physician. This is a small operation with a waiting room full of patients. Each of these clinicians has a job to do in the clinic dictated by clinic policy and state scope of practice law. The loss of any one clinician on this small team is a major disruption to the clinic wherein important organizational knowledge, workload distribution, and revenue generation are lost. Others must then pick up the slack which further exacerbates the workloads on other individuals and thus the cycle of burnout and overwork, repeats.
In large organizations, such as major health systems this small example is amplified in terms of scale and cost.
Automation and technological advancement have long been touted as a solution to labor challenges in both healthcare and other industries. This is a core opportunity for digital health. Automated check-in kiosks, patient portals, online scheduling software, text messaging, software billing automation, and other administrative automating technologies have done well in the clinic environment and have room to grow. When technology is truly able to replace or increase the productivity of a human worker, it has the potential to support practices in lessening the impact of the turnover “hamster wheel” and increasing patient throughput.
For example, an automated scheduling technology that ensures a high percentage of patient visits to provider availability is both a revenue optimizer and can reduce the need for human schedulers. If the scheduing department suffers from high turnover rates (which it often does), then more advanced scheduling technologies can make fewer humans more efficient thus reducing the impact of staff loss in addition to saving salary expense.
Administrative and operational digital health technologies have a strong value proposition to healthcare services organizations in that they can reduce labor costs, the disruption costs of turnover, and yield additional operational efficiencies by reducing frictions for patients and healthcare workers. In this way, and for this class of digital health companies, health services organizations operate much like other industries. They are on a quest to minimize operational costs and increase efficiency while growing market share and revenue. But, healthcare organizations are also on a quest for quality health outcomes which is more complex than concepts of product quality in other industries.
Clinical Digital Health Technologies and Workforce Issues
Administrative and operational digital health technologies are not often touted as the promise of digital transformation in healthcare services. They commonly fly under the radar. It is the clinical or medical technology that yields the most excitement, the most press, and the most opportunity to improve health outcomes—you know, the stuff that is important and makes us feel warm and fuzzy. In most cases, companies in this area will sell their products and services direct-to-consumer (e.g., Ro Health) or B2B2C (e.g., Virta, Avenue Health, Vivify). Clinical capability gains, remote monitoring, self-care support, digital therapeutics, novel treatment modalities, and digital diagnostic capabilities are the core of digital health. But, herein lies the core of the issue, these technologies often require significant operational realignments, reimbursement challenges and burdens, new workflows, learning curves, and specialized expertise for health services organizations. In short, humans have to do more things and decide to make a change.
I always tell my team that clinical digital health is a change management business. We think it’s about patients, and it is, but to our B2B healthcare customers, it’s about change. Here, even more importantly, it is all about clinicians and the workforce.
Take, for example, a software and hardware technology platform that allows asthma specialists to monitor inhaled corticosteroid adherence, the incidence of asthma attacks, the frequency of use of a rescue inhaler, and daily peak flow meter readings from a population of patients. Patients receive a mobile app that guides their treatment plan and provides education. The software provides daily information, alerts, and clinical decision support for the practice. This type of technology can improve health outcomes, prevent emergency department visits, reduce hospitalizations, and add peace of mind for patients. Read more about this here. There are also CPT billing codes that allow the clinic to pay for this technology and the resulting activities of proactive patient management—the RPM codes.
In the prior small practice example, the registered nurse may have been assigned to monitor the clinic’s patients on the monitoring program and perform outreach when necessary to help patients stay on track with their treatment plan. This new care model exists on top of traditional office visit operations at the clinic which are still occurring, thus, operationally, two unique business operating models are offered by the clinic. The traditional medical practice in which patients come for evaluation (Model 1) on a standard cadence and the “virtual clinic” where patients participate in a connected health model remotely (Model 2). The first model operates like any in-person service business whereas Model 2 is more like a call center.
Should the Model 2 nurse leave, the entire virtual clinic business model is disrupted and the monitoring burden is left to the folks operating the Model 1 clinic. The lost nurse was the subject matter expert on the technology, attended the training, and became the internal champion of the program. Finding, training, and hiring a replacement nurse for business Model 2 is a more difficult task than replacing a nurse for Model 1 as Model 2 is a novel concept and thus significant training will be essential for the replacement.
The clinic is turning a profit from Model 2, outcomes are good, and patients are satisfied with both the technology suite, which is purchased from a third-party company and the practice’s program. While turnover in Model 1 is still a challenge, turnover in Model 2 is more disruptive.
From the third-party digital health company’s perspective, the loss of that key nurse will impact user retention, user adoption, and program expansion. In some cases for a large book of business, turnover may result in the loss of accounts and churn.
Clinical digital health technologies are interesting to discuss as they have the potential to affect both workflows/operational costs and patient outcomes. Good products will address both. Software, as a medium, provides the opportunity to embed workflows into a new clinical intervention. Even among software companies focused only on providing relatively isolated, new clinical interventions (e.g., digital therapeutics) for a particular condition or care process, workflows are essential.
Clinical-focused Digital Health Technologies and Fee-For-Service Payment
New technologies that are designed primarily for clinical outcome improvement or treatment are often misaligned with fee-for-service payment models. FFS payment rewards efficiency and volume. Thus, change cost is incurred to adopt new treatment interventions as new technologies, products, and models often require reengineering of existing care processes.
Under FFS payment, the U.S. health system relies on the clinical professions and their organizations (e.g., the American Academy of Pediatrics) to set standards of care. After new technologies are developed and proven effective through controlled studies or real-world evidence, we expect that physicians, nurses, and other professors collectively work to implement them.
Pharmaceutical products are good examples to use. After a new drug is commercially available via significant regulatory processes, randomized clinical trials, and economic evaluations; the clinical specialty society responsible for the particular disease usually issues guidance based on the evidence. If the condition does not have a current treatment and the evidence is good, then the product will be adopted easily. If the product is competing with other existing standards of care, then the pathway to prescription will be more nuanced and complicated. The drug company will have to prove that their drug provides superior benefits to patients than the current standard, is useful after a current drug stops working, or benefits a particular subgroup of patients. This is a highly structured process that requires significant time and cost and the effectiveness of the new technology is ultimately a big factor in adoption.
Once approved for a particular indication, marketing activities commence. In pharmaceutical marketing, this usually involves major conference sponsorship, television advertisements, and a significant “ground game” (i.e., drug reps bringing lunch to every medical clinic in the country). The ground game is intended to educate prescribers on the value of the new drug and how to incorporate it into clinical practice. Fortunately for drug products, the workflow and workforce impacts on practices are low. Providers just write a prescription and the major logistics are managed by pharmacies, PBMs, and distributors. Pharma has long understood the need to make prescriptions as quick and easy as possible for clinicians as it determines future growth for a new drug.
Prescription digital therapeutics have tried to follow this route, but reimbursement, ordering logistics, and clinical adoption have been barriers. The promise of digital therapeutics as a business model leveraging the prescription drug-like infrastructure with SaaS-like cost of goods sold (COGS) and insurance reimbursement is attractive, but the system is not ready yet due to regulatory and payment policy hurdles (see Pear Therapeutics and Akili Interactive). One day, this will be feasible and attractive given the right clinical evidence, the right market conditions, and a clear reimbursement pathway both federally and commercially. Until then, many digital therapeutics companies have pivoted to direct-to-consumer models and B2B2C approaches.
Digital health technologies that focus on B2B or B2B2C models such as those for chronic care management, remote patient monitoring, and telehealth are particularly sensitive to the change management and workforce pressures. Other technology concepts sensitive to workforce factors in digital health include software-integrated medical devices, digital diagnostics, and clinical decision support software. These products are becoming increasingly common in niche specialty conditions (e.g., multiple sclerosis) in addition to broader market models (e.g., Teladoc).
For example, several hardware-software digital health companies are developing software-enabled technologies and medical devices specifically for multiple sclerosis and Parkinson’s Disease. These companies are looking to enhance assessment and continuous monitoring of these conditions to help physicians better manage dosing and treatment plans by integrating hardware and software into the clinic environment.
Just like the asthma clinic example above, these technologies and their developers are subject to the humans that operate clinics caring for their target patient populations. For these types of companies, how the technologies impact humans is equally critical as clinical evidence and professional adoption. These companies must both prove treatment effectiveness and operational feasibility to their target customers—often a higher burden than the drug pathway.
In the FFS payment environment, the adoption of a novel, in-clinic Parkinson’s software platform and corresponding medical device may be attractive clinically, but unattractive operationally and from a business perspective. Even if the technology has reimbursement potential, the magnitude of the payment and relative value to other existing clinical services dictates the level of alignment with business needs. This is the core of the argument of this article.
The impact on clinicians and the workforce is a critical area for digital health companies to address first in product design and then in sales, marketing, and implementation. Your technology or clinical intervention might be the “greatest solution ever” to improve health, but if it is operationally or financially burdensome then you will have challenges with uptake. Many digital health technologies are not reimbursed directly as is the case with drugs, so the indirect reimbursement via a service-driven CPT code further impacts the need for careful alignment in these areas.
Clinical-focused Digital Health Technologies and the Value-based Payment Environment
In the U.S. healthcare market, the move toward value-based payment (VBP) is positive for digital health companies. A significant number of Medicare beneficiaries and other covered populations are covered under some form of VBC model. For example, in the Medicare Shared Savings Program (MSSP), one of several accountable care organization (ACO) models, care delivery organizations are incentivized to provide better care due to incentives tied to quality measures and incentives to reduce a patient’s total cost of care (e.g., brand name drugs, emergency visits, and hospitalizations).
For the asthma connected-care model example above, the practice is paid FFS for the remote monitoring services, but under an MSSP-like ACO model, if the asthma RPM program prevents ED visits and hospitalizations then the practice will share in the savings that result to the payor. Thus, the clinic has further incentives to adopt the RPM-based connected care model due to business incentives over and above the standard FFS reimbursement.
Under this same type of model, quality metrics are also used to provide incentives for desirable outcomes like medication adherence. If the RPM-based asthma care model helps patients stay adherent to inhaled corticosteroids and increases the adherence rate across the practice patient population, then that practice may receive bonus payments for improvement in the corresponding HEDIS measure.
Thus, for digital health companies focused primarily on clinical capability gains there is a growing incentive structure in the healthcare services market to facilitate adoption if FFS incentives have been prohibitive, but ROI must be established using a cost-effectiveness framework, first.
A Model for Evaluating Digital Health Innovations
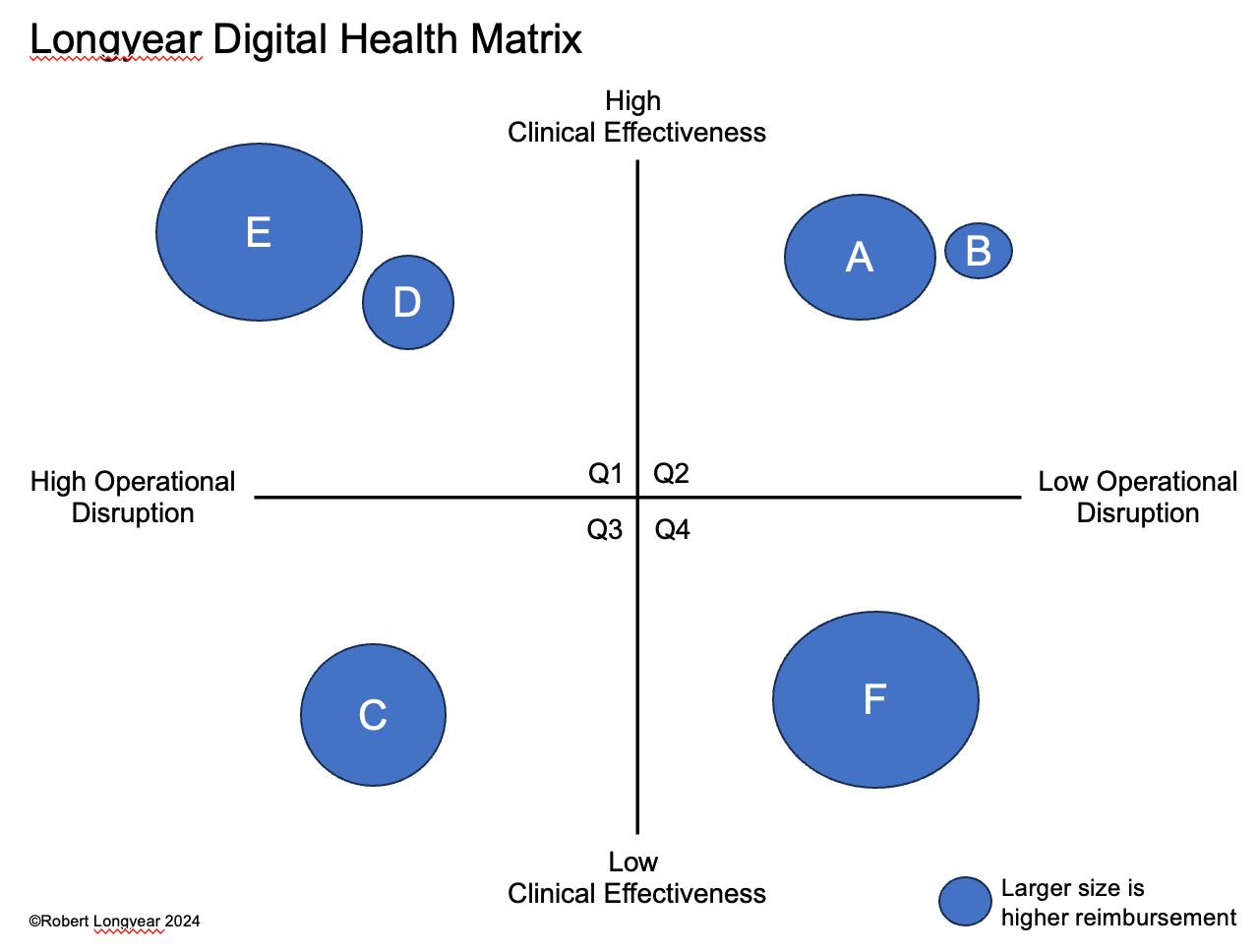
A useful model for evaluating the success of innovations in digital health is outlined, below (Figure 1). In the model, the level of operational disruption is plotted against the demonstrated (or, perceived) clinical effectiveness of the technology or care model. The size of the bubble for that particular technology or care model corresponds with the magnitude of reimbursement (or, ROI for a clinic). I outline the characteristics of each new technology, below.
Figure 1
Technology A
Technology A causes low operational disruption and low workforce disruption with high clinical effectiveness. Compared to technology B, its reimbursement is relatively higher. This technology is the most attractive one on the matrix. Technology A could be thought of as a pharmaceutical product like Ozempic. It is clinically effective and causes low operational disruption in clinics via the drug prescription pathway. Some digital health technologies may begin to resemble Technology A as digital therapeutics navigate the regulatory environment and logistics become less burdensome on clinics. Certain digital diagnostics with favorable reimbursement, low operational disruption, and good effectiveness are becoming more common.
Technology B
Technology B is similar to Technology A with even lower operational disruption and high clinical effectiveness, but comparably low reimbursement. Thus, its adoption rate will hinge on the economics determined by the magnitude of the rate. Unless professional societies suggest the technology as a standard of care, this type of innovation may see slower uptake than one with similar characteristics with higher reimbursement. Professional society recommendations can allow companies in this situation to seek higher reimbursement, especially if cost-effectiveness research shows favorable results. In this case, payors may see ROI if covered as well. The bili-hut is a good example of this type of technology where reimbursement is relatively low, but the other factors are favorable. For the company, low COGs can still yield positive business outcomes.
Technology C
Technology C is low in clinical effectiveness and high in operational disruption but yields reasonable and moderate reimbursement relative to other concepts in the model. This is like a connected medical device that marginally improves outcomes over existing technologies and requires a good deal of training, workflow adjustments, and coordination to implement. This is like adding an activity sensor to a knee replacement device and expecting the surgeon to perform long-term monitoring of activity. Surgical practices are not set up for chronic disease care and thus this represents a significant workflow disruptor with limited clinical value. This might add some value in the post-surgical period, but, at a certain point, activity monitoring on a long-term basis is more valuable to a primary care physician for other conditions.
The RPM code set has set off a race among “dumb” medical device manufacturers to create “smart” devices some of which are of limited clinical value and that, despite good RPM reimbursement, just do not fit into operations well.
Technology D
Technology D has high clinical effectiveness and high operational disruption with relatively low reimbursement compared to technology E in the same quadrant. Technology D, for example, could be the asthma-connected care model described above. If both Technology D and Technology E are useful to asthma care specialists, then there is a problem. Technology D is less disruptive than Technology E, but the relatively lower reimbursement means that it competes for the limited human capital resources and attention of the asthma specialist market. Technology D can compete better with Technology E by becoming less disruptive via product enhancements, better software integration, and other support models that address the human factors involved in program development and sustainment.
Technology E
Technology E could be a software + cell therapy company that helps patients and clinics navigate the process of cell therapy treatment. For the sake of discussion, let’s say this treatment targets asthma patients and can also reduce ED visits and hospitalizations, just like Technology D, because it works to prevent asthma attacks. Reimbursement for the clinic in this case may be quite significant, but workflow disruptions are also large. Thus, technology D and E are useful examples of innovations seeking to affect the same outcomes, but with differences in the key variables tracked in this model. The model can be used to look at products and innovations that compete or across different concepts and indications.
Technology F
Technology F is low in clinical operation disruption and low in clinical effectiveness. Shown here with large reimbursement, the best example of this in the real world is the recent approval of Alzheimer’s drug, Aduhelm. Clinically, there are questions about the clinical value of the drug despite its approval. Operationally, this drug is accessible via standard specialty pharmacy pathways with a simple prescription from a provider. Reimbursement is quite high and due to the lack of another treatment option, it will likely gain adoption.
In digital health, the Technology F example may be a digital therapeutic that has demonstrated statistically significant effects on depressive symptoms, but with a low effect size compared to pharmacologic treatment. In this case, the company allows the app to be downloaded via an app store without a prescription from a provider. Providers may recommend the application to patients and thus patients have ease of access, and direct cash pay ability yielding favorable payment to the company. In the future, this scenario may play out as digital therapeutics gain reimbursement that may be crosswalked to existing medical devices and drug rates.
Characteristics of the Quadrants
This concept model does not account for all factors that determine the success of digital health products and services but provides an important framework for how healthcare delivery organizations think about adoption.
Quadrant 2 (Q2) with high reimbursement will be the most successful, but even lower reimbursement can still lead to rapid adoption. Quadrant 3 (Q3) is not likely to produce success. Identifying Q3 companies is important. Quadrant 1 (Q1) relies on high-reimbursement and high-quality evidence to garner adoption but may take more time than Quadrant 2, thus companies in this quadrant must raise additional funds to continue operations over time until operational disruption can be addressed. Quadrant 4 (Q4) is very beholden to the magnitude of reimbursement. Given the low operational disruption, the company’s success will be determined by COGS and reimbursement rate. In a Q4 situation, effective marketing can yield results despite low clinical effectiveness, particularly if the product experience is good.
Conclusion
The impact of healthcare products and services on the humans that administer and recommend them is important to identify in the current healthcare environment. Given high rates of turnover and workforce dropout, digital health companies must understand their strategy to ensure that workforce risk is minimized in product design, contracting, implementation, and ongoing operations.